367-25-9
- Product Name:2,4-Difluoroaniline
- Molecular Formula:C6H5F2N
- Purity:99%
- Molecular Weight:129.109
Product Details;
CasNo: 367-25-9
Molecular Formula: C6H5F2N
Appearance: dark red liquid
Top Purity Chinese Factory Supply 2,4-Difluoroaniline 367-25-9 Cheap Price
- Molecular Formula:C6H5F2N
- Molecular Weight:129.109
- Appearance/Colour:dark red liquid
- Vapor Pressure:3.8mmHg at 25°C
- Melting Point:-7.5 °C(lit.)
- Refractive Index:n20/D 1.506(lit.)
- Boiling Point:168.4 °C at 760 mmHg
- PKA:3.26±0.10(Predicted)
- Flash Point:62.8 °C
- PSA:26.02000
- Density:1.289 g/cm3
- LogP:2.12820
2,4-Difluoroaniline(Cas 367-25-9) Usage
|
Description |
2,4-difluoroaniline is an important intermediate used in organic synthesis, pharmaceuticals agrochemicals, dyes, and agricultural chemicals. |
|
Reference |
R. Garth Pews, J. A. Gall, Process and intermediates for the preparation of 2,6-difluoroaniline, Paten US5041674A |
|
Chemical Properties |
dark red liquid |
|
Uses |
2,4-Difluoroaniline is an important raw material and intermediate used in organic synthesis, pharmaceuticals agrochemicals and dyestuff fields. |
|
General Description |
Dark reddish-purple liquid. |
|
Air & Water Reactions |
2,4-Difluoroaniline may be sensitive to prolonged exposure to air. . Water soluble. |
|
Reactivity Profile |
Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. |
|
Health Hazard |
ACUTE/CHRONIC HAZARDS: 2,4-Difluoroaniline may be irritating to tissues. |
|
Fire Hazard |
2,4-Difluoroaniline is combustible. |
InChI:InChI=1/C6H5F2N/c7-4-1-2-6(9)5(8)3-4/h1-3H,9H2
367-25-9 Relevant articles
Synthesis of carbon-supported Pd/SnO2 catalyst for highly selective hydrogenation of 2,4-difluoronitrobenzene
Zhao, Jia,Ma, Lei,Xu, Xiao-Liang,Feng, Feng,Li, Xiao-Nian
, p. 1137 - 1140 (2014)
Halogenated anilines have a wide range o...
Efficient and recyclable bimetallic Co–Cu catalysts for selective hydrogenation of halogenated nitroarenes
Lu, Xionggang,Ren, Jiaan,Sheng, Yao,Wang, Xueguang,Wu, Baoqin,Zou, Xiujing
, (2021/12/20)
Silica supported N-doped carbon layers e...
Novel preparation method of 2, 4, 5-trifluorophenylacetic acid
-
Paragraph 0037; 0075; 0080-0081; 0097-0100; 0102-0103; ..., (2021/06/23)
The invention discloses a novel preparat...
Efficient synthesis method of meta-fluoranisole (by machine translation)
-
Paragraph 0026; 0033; 0036; 0043; 0046; 0053; 0056; 0063, (2020/06/05)
The method is characterized by comprisin...
Highly selective hydrogenation of halogenated nitroarenes over Ru/CN nanocomposites by: In situ pyrolysis
Yue, Shengnan,Wang, Xueguang,Li, Shaoting,Sheng, Yao,Zou, Xiujing,Lu, Xionggang,Zhang, Chunlei
, p. 11861 - 11869 (2020/07/28)
A highly chemoselective and recyclable r...
367-25-9 Process route
-
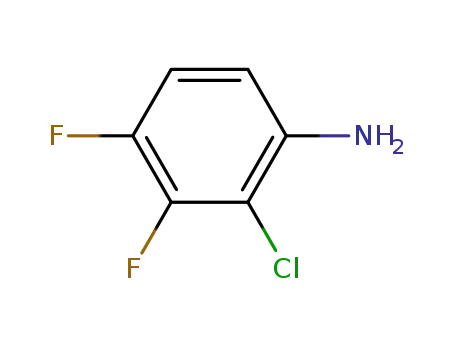
- 36556-48-6
2-Chloro-3,4-difluoroaniline

-
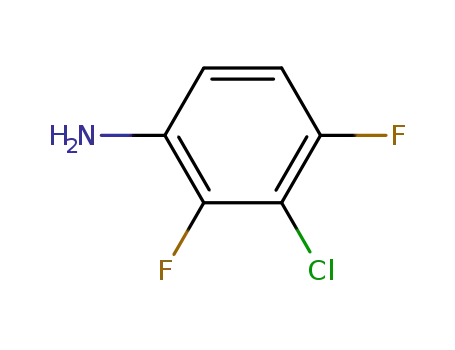
- 2613-34-5
2,4-difluoro-3-chloro-aniline

-
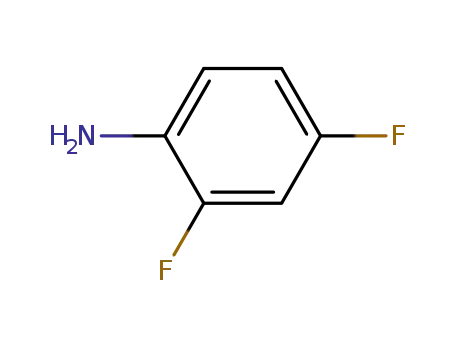
- 367-25-9,76563-56-9
2,4-difluorophenylamine
| Conditions | Yield |
|---|---|
|
With sodium hydroxide; palladium; In hydrogenchloride; methanol; water;
|
99% |
-
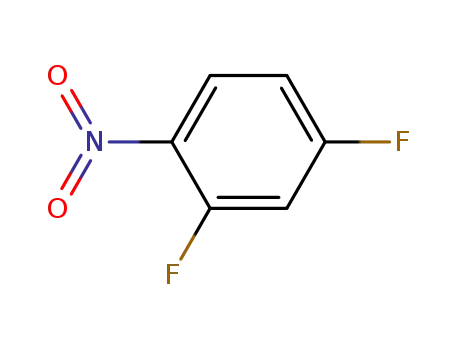
- 446-35-5
2,4-Difluoronitrobenzene

-

- 367-25-9,76563-56-9
2,4-difluorophenylamine
| Conditions | Yield |
|---|---|
|
With hydrazine hydrate; at 90 ℃; for 1.33333h; chemoselective reaction;
|
99% |
|
With sodium hypophosphite; 1% platinum on charcoal; hydrogen; at 70 - 90 ℃; for 2.75h; under 6750.68 Torr; Reagent/catalyst; Temperature; Pressure; Autoclave; Inert atmosphere;
|
98.6% |
|
With hydrazine hydrate; at 100 ℃; for 1.33333h;
|
95% |
|
With hydrogen; at 20 ℃; Autoclave;
|
95% |
|
With hydrogenchloride; tin(ll) chloride;
|
|
|
With hydrogenchloride; iron;
|
|
|
With sulfuric acid; iron;
|
|
|
With iron; ammonium chloride;
|
|
|
platinum;
|
|
|
With hydrogen; In methanol; at 20 ℃; for 1h; under 750.075 Torr; chemoselective reaction;
|
|
|
With hydrogen; In methanol; at 79.84 ℃; under 7500.75 Torr; Autoclave;
|
|
|
With hydrogen; In ethanol; at 89.84 ℃; under 7500.75 Torr; Reagent/catalyst; Autoclave;
|
|
|
With palladium on activated charcoal; hydrogen; In methanol; at 20 ℃;
|
810 mg |
|
With hydrido(tetrahydroborato-HH'){1,1,1-tris(diphenylphosphinomethyl)ethane-PP'P''}iron; In tetrahydrofuran; at 80 ℃; for 24h; Inert atmosphere;
|
|
|
With hydrogen; In water; at 60 ℃; Large scale;
|
3306 kg |
|
With hydrogen; In ethanol; at 80 ℃; for 1.91667h; under 15001.5 Torr; chemoselective reaction; Catalytic behavior;
|
|
|
With hydrogen; In ethanol; at 120 ℃; for 3h; under 15001.5 Torr; Autoclave;
|
367-25-9 Upstream products
-
446-35-5

2,4-Difluoronitrobenzene
-
1435-43-4
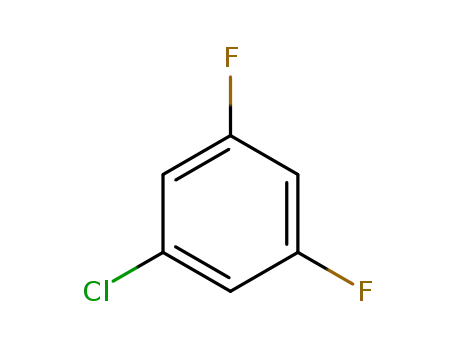
1-chloro-3,5-difluorobenzene
-
1481-68-1
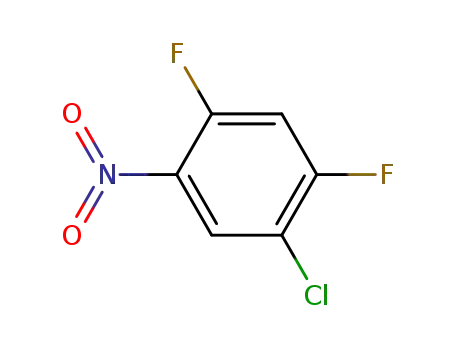
5-chloro-2,4-difluoronitrobenzene
-
83164-33-4
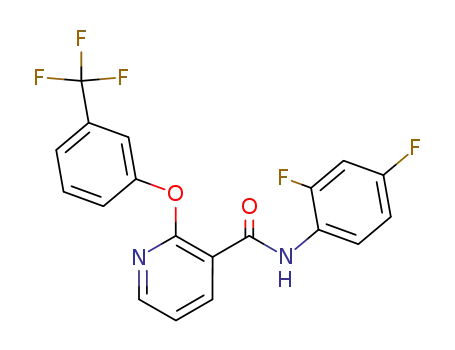
N-(2,4-difluorophenyl)-2-(3-trifluoromethylphenoxy)-3-pyridinecarboxamide
367-25-9 Downstream products
-
1581-06-2
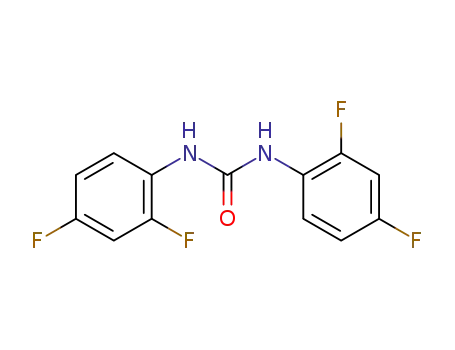
2.2'.4.4'-Tetrafluor-diphenyl-harnstoff
-
366-46-1

hydroxyimino-acetic acid-(2,4-difluoro-anilide)
-
367-27-1
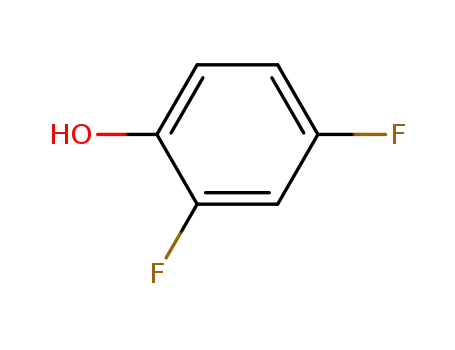
2,4-difluorophenol
-
2265-93-2
2,4-difluoro-1-iodobenzene
Relevant Products
-
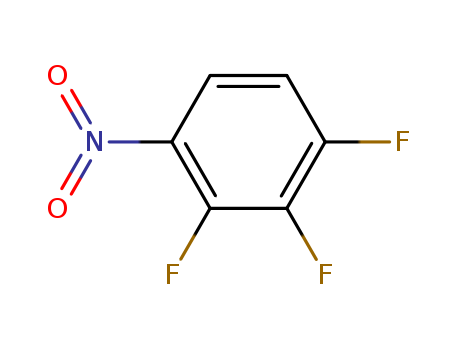
1,2,3-Trifluoro-4-nitrobenzene
CAS:771-69-7
-
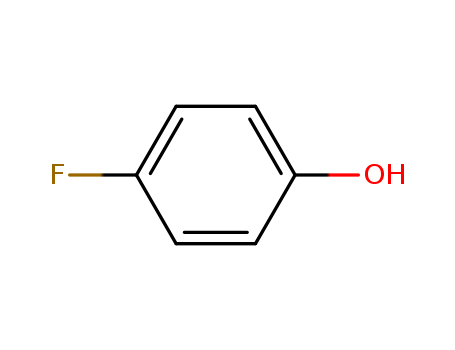
4-Fluorophenol
CAS:371-41-5
-
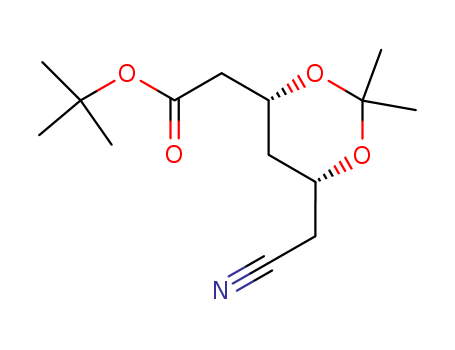
Atorvastatin intermediatesAts-8
CAS:125971-94-0